SRT (Steriotactic Radiosurgery)
End-To-End Phantoms
LIQUIPHIL™ END-TO-END PHANTOM
The LiquiPhil™ End-to-End Phantoms were developed to provide stereotactic localization and dose verification for radiosurgery machines. The phantoms may be used for a variety of radiation therapy applications including periodic quality assurance evaluations and acceptance testing and end-to-end testing in conformance to AAPM TG-54. The phantoms may also be used to perform re-evaluations after equipment or software upgrades.
Charles W. Coffey, II, Ph.D., established the design criteria that The Phantom Laboratory used in constructing the LiquiPhil™ Head Phantom and LiquiPhil™ Pelvis Phantom. In 2013 The Phantom Laboratory worked with Michael S. Gossman, M.S., DABR, FAAPM, to develop the LiquiPhil™ Head Phantom with Bite Feature, providing for both stereotactic radiosurgery and radiotherapy quality assurance with bite tray systems.
For detailed information please refer to the LiquiPhil™ Head End-to-End Phantom and Data Sheet, the LiquiPhil™ Head Phantom with Bite Feature and Data Sheet (Bite Feature) or the LiquiPhil™ Pelvis Phantom Manual and Data Sheet.
Available END-TO-END Models
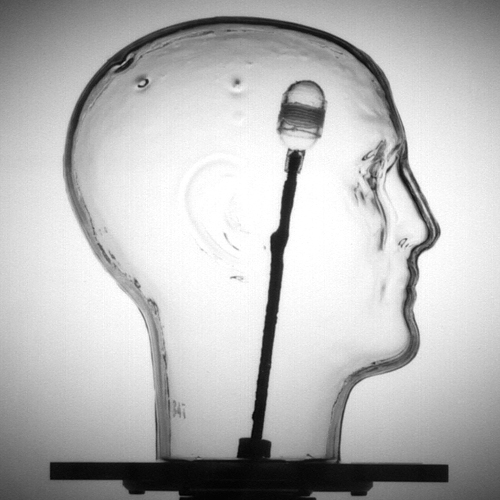
LiquiPhil™ Head Phantom: A water filled head with a repositionable tumor vessel that can be adapted to accommodate ionization chambers, TLDs, film or dosimetric gels. Ideal for testing stereotactic treatment using halos, and surface monitoring positioning systems.
LiquiPhil™ Head Phantom with Bite Feature: This model is similar to the LiquiPhil™ Head Phantom with the addition of a bite feature for use with immobilization bite tray systems.
LiquiPhil™ Pelvis Phantom: A life size pelvic form with with a repositionable tumor vessel that can be adapted to accommodate ionization chambers, TLDs, film or dosimetric gels. Ideal for pre-treatment verification for both conventional and IMRT plans.
Design
The phantoms’ design provides full simulation of the localization and irradiation sequences. The anatomically accurate forms are filled with water to simulate the radiation absorption and scatter of human soft tissue. The heavy-duty outer shell of the LiquiPhil™ Phantom is designed to accommodate the anchoring screws needed for proper positioning head frames. An internal container called a tumor vessel can be positioned anywhere within the head form by manipulating an external position rod. This vessel may be filled with either a radiation-sensitive gel for alignment evaluations or with radiation dosimeters for quantitative dose measurements. The LiquiPhil™ Head Phantom with Bite Feature features a movable lower jaw and upper teeth provide for the use of bite tray systems.
For conventional and IMRT dose distribution and verification the LiquiPhil™ Pelvis Phantom is an excellent tool for final quality verification of therapy dose delivery and for comparing the delivered dose profiles for different treatment plans.
Stereotactic Localization
The phantom provides an accurate evaluation of stereotactic localization. To perform an evaluation dose sensitive gels, non light sensitive film, TLDs, or small ion chambers are used. The tumor vessel or chamber is positioned within the phantom’s head form and the radiosurgery head frame is attached to the phantom. Standard protocols are followed to locate the x, y and z coordinates of the patient’s tumor through CT or MR imaging. The phantom is then mounted in the radiosurgery system and irradiated. Once the irradiation has been completed the tumor vessel is removed from the phantom head form and dose or dose profile is evaluated.
Phantom Materials
The shell of a LiquiPhil™ Phantom is formed from ¼” cellulose acetate butyrate sheet, a transparent material chosen for its strength and low water absorption. The shell is mounted on a polycarbonate end plate. The tumor port and cover plate assembly are attached with nylon screws to the end plate. The cover plate is removable for internal access. Before phantoms are shipped to customers all of their seams are tested to ensure a water-tight seal. The Phantom Laboratory provides a wooden storage case for each LiquiPhil™ Phantom. For the LiquiPhil™ Head Phantom with Bite Feature, the upper teeth are cast from a bone-simulating urethane and the lower jaw is cast from a clear urethane.
Tumor Assembly
The tumor vessels are molded out of Barex®, an oxygen impermeable material that is ideal for use with radiation-sensitive gels.
Three tumor vessels are included with the LiquiPhil™ Head Phantom, with one vessel being used in the phantom at a time. The head phantom vessels has an inside length of 4cm and inside diameter of 2cm, and a radius of 1cm at each end. The pelvis tumor vessel has an inside diameter of 10cm and an inside length of 12cm.
The two ends of the tumor vessel are threaded together at the center to allow easy access into the vessel. The user may position the tumor vessel anywhere within the head form by manipulating the phantom’s external position rod. After the desired position is reached, the cylinder is locked into place by hand-tightening a lock nut on the rotation ball and a lock bolt on the position rod.
Optional Equipment
The optional TLP211 plug holder, which is mounted into the tumor vessel, is designed to hold 5mm plugs for TLDs.
The optional TLP212 and TLP213 film holders, which are mounted into the tumor vessel, hold dosimetry film securely in place.
Specially ordered commercially available chambers and scintillation detectors may also be used in the LiquiPhil™ Phantom. Contact The Phantom Laboratory for current information on these devices.